Prescription opioid abuse is a significant public health concern in the United States, particularly abuse of controlled-release formulations that contain higher opioid content.1 The enormous abuse potential of prescription opioids is now well-recognized, and strategies to mitigate these risks are being implemented.1,2 This article provides an overview of the government’s position on the issue of prescription opioid abuse; the guidance provided by the US Food and Drug Administration (FDA) for development of abuse-deterrent formulations (ADFs) of opioids, including the requirement for postmarketing population-based health studies to confirm the benefit of these agents; and the outcomes of those postmarketing studies.
Current FDA Guidance
Recognizing the magnitude of the prescription opioid abuse, misuse, and diversion issues in the United States, in 2011, the Office of National Drug Control Policy (ONDCP) released a national action plan with the goal of reducing nonmedical use of prescription drugs by a balanced policy that includes action in 4 major areas: (1) education, (2) monitoring, (3) proper disposal, and (4) enforcement.1 Toward that end, prescription monitoring, overdose prevention, drug take-back, and educational programs have been initiated.1,3 In addition, the FDA required manufacturers to develop Risk Evaluation and Mitigation Strategy (REMS) programs for controlled-release prescription opioids, under which they provide educational materials to prescribers and patients to promote appropriate opioid prescribing, disposal, storage, and safe use. Importantly, the ONDCP’s plan emphasized the need for research initiatives to develop pain treatment strategies with no abuse potential as well as ADFs of opioids and other drugs with abuse potential.1 In addition, the plan called for advancement in the design and evaluation of epidemiologic studies to address changing patterns of abuse, and it recommended that the FDA provide guidance to the pharmaceutical industry on development of ADFs and on postmarketing assessment of their performance.
In response to this call for action, the FDA published draft guidance for premarketing and postmarketing assessments of the abuse liability of prescription opioids, while acknowledging that “the science of abuse deterrence is relatively new, and both the formulation technologies and the analytical, clinical, and statistical methods for evaluating those technologies are rapidly evolving.2” The guidance outlines the levels of evidence required to demonstrate the abuse liability of ADFs of prescription opioids at the premarketing and postmarketing stages (Figure).2
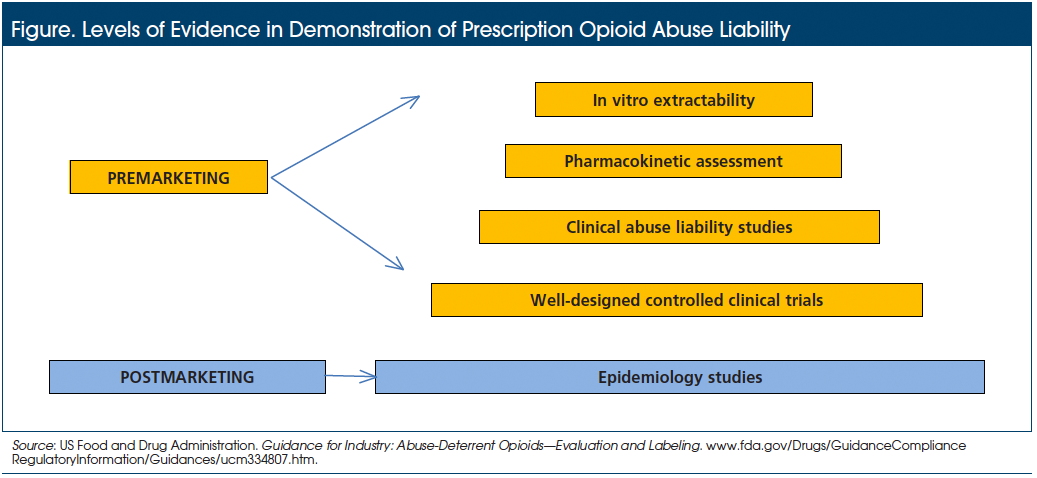
Guidance for Premarketing Studies of Opioid ADFs
Premarketing studies are primarily expected to characterize the abuse-resistance properties of a product under controlled conditions, in contrast to postmarketing studies, which determine whether the marketed ADF results in meaningful decreases of prescription opioid abuse in real-world settings.2 For premarketing trials, the FDA guidance calls for 3 categories of studies to obtain a full and scientifically rigorous understanding of the impact of the investigational product on opioid abuse: (1) laboratory-based in vitro manipulation and extraction studies, (2) pharmacokinetic assessments, and (3) clinical abuse potential evaluations.
In vitro studies are required to include assessments of various physical and chemical manipulations of the drug, such as evaluating the ease with which the potentially abuse-resistant properties of the ADF can be overcome for purposes of abuse by different routes of administration, the ease with which an immediate-release formulation may be prepared for alternate routes of administration, or the ease with which the opioid antagonist, if present, may be separated from the opioid agonist with the intent of circumventing the product’s abuse-deterrent properties.2 Specifically, the ability to crush, cut, grate, or grind the formulation using readily available items such as spoons, cutters, and coffee grinders should be assessed under heat and cold conditions, with a particular focus on the particle size distribution, since it directly corresponds to the rate of opioid extraction. In addition, extractability and solubility studies using a variety of commonly available solvents, such as water, vinegar, and ethanol, are required to be performed to determine the ease of evading the drug’s abuse-deterrent properties or extracting opioid. The guidance stresses the importance of being cognizant of the common routes of administration of individual drugs when designing the evaluation studies, such as determining the opioid concentration in a small injection volume and viscosity of fluid for a product with potential for intravenous injection.
The guidance states that the purpose of pharmacokinetic studies is to compare the pharmacokinetic profiles of the manipulated formulation with the original, as well as those of appropriate comparator drugs, through 1 or more routes of administration.2 Relevant pharmacokinetic parameters to measure include maximum concentration, time to maximum concentration, area under the curve, and terminal elimination half-life, with and without exposure to food and alcohol.
The goal of clinical studies of abuse liability is to assess the impact of the ADF on the attractiveness or likability of the formulation to abusers, preferably in a double-blind, randomized, crossover trial that makes use of a positive comparator and a placebo control.2
Guidance for Postmarketing Epidemiologic Studies of Opioid ADFs
Although best practices for postmarketing epidemiologic studies have not been established,2 the FDA has outlined a few characteristics that formal studies should have:
- Use outcomes that provide meaningful measures of abuse deterrence
- Produce estimates of abuse deterrence that are nationally representative or are based on data from large geographic regions
- Assess overall and route-specific abuse and abuse deterrence
- Be sufficiently powered to assess meaningful changes in drug abuse.
Overall, it is expected that the studies will detect a change in the occurrence of abuse and abuse-related clinical outcomes as a result of the ADF. The FDA guidance specifies that the study cohort should include a high-risk population, such as one of known drug abusers, and outcomes of reported abuse should include prevalence and frequency of abuse, such as days of abuse in the past 30 days, whereas clinical outcomes should include prevalence or rates of overdoses, poisonings, addiction, and death. Study designs may be varied, including the use of claims data, the use of surveillance systems, and observational studies.
Epidemiologic Assessments of Opioid ADFs
Over the past decade, highly encouraged by regulatory agencies, manufacturers of opioids have been actively involved in the development of opioid formulations with new physiochemical barriers to deter abuse.3-5 Several ADFs of opioids are currently available, including a reformulated extended-release (ER) opioid with a polymer coating that is designed to render it crush-resistant, 2 reformulated opioids with a polyethylene oxide matrix that is designed to be crush-resistant, and combinations that contain opioid antagonists, which are released only upon product manipulation, to block the euphoric effects of opioids.
In vitro and clinical assessments of these ADFs have demonstrated their abuse-resistance properties (reviewed elsewhere in this series).3-5 In an effort to extend these studies and to fulfill the FDA mandate, several postmarketing studies of ADF opioids have been conducted. Investigators in these studies have sought to examine the effects of ADFs on abuse-related outcomes in the real-world setting.3-5 This large body of accumulated pharmacoepidemiologic evidence attests to the abuse-deterrent effects of opioid ADFs.
The reformulated version of a semisynthetic ER opioid was the subject of many studies following its approval in August 2010.6-11 These studies reported reductions in abuse of this drug assessed in real-world settings such as in substance abuse treatment centers. Cicero and colleagues reported results of sequential self-administered surveys completed by 2566 patients with diagnosed opioid dependence/addiction who were entering treatment programs in the United States and for whom a prescription opioid was the primary drug of abuse.6 After the reformulation of this ER opioid was approved, there was a significant reduction in the proportion of patients using this agent as the primary drug of abuse, from 35.6% to 12.8% (P <.001). This was also evidenced by significant reductions in the use of this agent to “get high at least once in the last 30 days,” from 47.4% to 30.0% (P <.001). Interestingly, a concomitant increase was reported in the abuse of other opioids, from 20.1% to 32.3% (P = .005), whereas heroin use rose from approximately 10% to 20%, suggesting that patients preferred heroin or other prescription opioids over the reformulated ER opioid.
Larger studies have since confirmed these results.2-5 Butler and colleagues reported results of a sentinel surveillance sample of 140,496 patients assessed for substance abuse treatment at 357 US centers between June 1, 2009, and March 31, 2012.7 This study assessed the prevalence of past-30-day abuse via any route, as well as abuse through oral, nonoral, and specific routes of administration, for the non-abuse-resistant ER formulation and comparators before and after introduction of the reformulated version. Findings of this large study showed that overall abuse of the reformulated drug was 41% lower than historical abuse of the non-abuse-resistant version (P <.0001). Oral abuse was 17% lower with the reformulated version than with the original formulation, and nonoral abuse was 66% lower (P <.0001 for both comparisons). In contrast, significant reductions in abuse were not observed for the comparators (other opioids).
Cassidy and colleagues explored temporal trends in the prevalence of prescription opioid abuse in a sentinel sample of 232,874 adults at 437 facilities who were being assessed for or entering substance abuse treatment.8 The researchers used a proprietary system for monitoring prescription drug use to examine changes in abuse patterns before and after introduction of an ADF for an ER opioid. Over the 4-year study period (January 1, 2008-December 31, 2011), increases in abuse prevalence occurred overall for all prescription opioids, including for ER opioids. However, there were differences in the prevalence of abuse of different prescription opioids, suggesting that switching of opioid compounds, possibly as an alternative for abuse, occurred after ADF introduction. These changes in abuse during the post-ADF period did not extend to illegal drugs, as evidenced by declining trends in the abuse of heroin and cocaine over time.3
Coplan and colleagues reported results from a population-based study that used the National Poison Data System, maintained by the American Association of Poison Control Centers, to assess the effects of a reformulated ER opioid on abuse outcomes.9 The researchers analyzed calls to poison control centers regarding the original version of the opioid during the 1-year period before introduction of the reformulation (July 2009-June 2010) versus the 2 years afterward (September 2010-September 2012), and then compared the results with trends for other single-entity similar opioid tablets and heroin. The number of calls that were related to the original ER opioid decreased steadily after the reformulation was introduced; striking decreases occurred overall and in calls about suicide, therapeutic errors, accidental ingestion, and adverse reactions. Overall, the results indicated that reformulation of the ER opioid tablets was associated with a 36% reduction in abuse, whereas abuse of other single-entity opioid tablets and heroin increased by 21% and 35%, respectively. Similarly, therapeutic errors, defined as “unintentional deviation from a proper therapeutic regimen that results in wrong dose,” decreased by 20% with the reformulated version, indicating that reformulation reduces accidental exposure to potentially fatal doses from tablets that may be crushed, chewed, or dissolved by patients or healthcare providers for ease of ingestion. In addition, accidental exposures, including unintentional ingestion by children gaining access to these substances, decreased by 39% overall, and accidental exposure among children aged 1 to 2½ years decreased by 51% postreformulation.4
Severtson and colleagues reported declines in abuse, therapeutic errors, and diversion into illegal markets following the introduction of a reformulated ER opioid.10 At the Rocky Mountain Poison and Drug Center, in Denver, CO, data for the 2-year period prior to introduction of the ADF (October 2008-September 2010) were compared with data for the 1.5 years after introduction (October 2010 through March 2012). Following the introduction of the ADF, calls related to abuse declined by 38% per population and by 32% per unique recipient of dispensed drug. Calls related to therapeutic errors were reduced by 24%, whereas diversion reports declined by 53%. These declines were greater than declines observed for all other prescription opioids combined. Moreover, the street price of the reformulated ER opioid was found to be significantly lower than that of the original formulation, which may be considered a surrogate measure of its attractiveness in terms of abuse potential.
In rural Appalachia, the prevalence of abuse of an original version of an ER opioid declined substantially following the introduction of the reformulated version, and abuse rates of the reformulated version remained low.11 Between December 2010 and September 2011, Havens and colleagues conducted structured interviews in that region with 189 individuals who had a history of abusing an ER opioid. One-third of respondents said that in the past 30 days, they had abused the reformulated version through any route (average, 1.9 days/month), whereas 96% had abused the original version (19.5 days/month). Rates of abuse of the ADF were particularly low via snorting (5% vs 70% for original opioid) and injection (0.5% vs 51% for original opioid).
More recently, an observational study examined whether patients with pain avoid switching to ADFs—and the extent to which those patients are likely to be prescription drug abusers.12 Michna and colleagues analyzed proprietary pharmacy and medical claims data following the introduction of the reformulated versions of 2 ER opioids in patients who had used the original non–abuse-resistant formulations of these drugs for at least 6 months before reformulation.12 The results showed that 31% to 50% of patients avoided switching to reformulated ER opioids, preferring to switch to non-ADF opioids or to discontinue opioid use altogether. Moreover, the patients who avoided switching to ADFs showed higher rates of diagnosed opioid abuse than those who transitioned to the reformulated ER opioids.
Systematic Internet surveillance studies have emerged as a feasible and important means to monitor and evaluate the overall impact of ADFs with regard to their abuse potential and attractiveness.13,14 Because Internet posts are uncensored, peer-to-peer communications, it is believed that they may provide valuable insight into ongoing efforts to undermine the abuse-deterrent properties of ADFs, as well as reveal any practical abuse methods that have been designed by abusers. While monitoring over 45,000 Internet posts related to opioids over a study period of August 2010 through September 2013, McNaughton and colleagues determined that only a relatively small proportion reported abuse of the reformulated version, particularly via injection or snorting routes, and abuse decreased over time.14
Conclusions
The accumulated evidence from postmarketing epidemiologic studies of opioid ADFs points to declines not only in ADF abuse patterns, but also in therapeutic errors, accidental exposures, and diversion of prescription opioids. Such data serve to provide a comprehensive understanding of the public health impact of reformulation on nonmedical use of prescription opioids, individually and as a group. The evidence thus far indicates that reformulating abused prescription opioids to include abuse-resistant properties may be an effective approach to reduce abuse of these medications.
However, a major observation from postmarketing epidemiology studies of a reformulated version of an ER opioid was that the introduction of the ADF was associated with increased abuse of other opioids and illicit drugs. Therefore, a reasonable inference may be that the drug is being replaced with other opioids and agents that are more amenable to manipulation. Encouragingly, it appears that novel methods to bypass the abuse-resistant properties of reformulated drugs have not been deduced at the present time, as evidenced from Internet surveillance data as well as reported decreased rates of abuse, misuse, and accidental exposure associated with reformulated opioids, strongly supporting the effectiveness of the ADF approach. Taking these findings together, it may be concluded that the current ADF methodologies alone will not likely be adequate to curb nonmedical opioid use; however, they may be effective as part of a comprehensive effort that includes other interventional strategies such as REMS programs, state prescription monitoring, and overdose prevention programs.
References
- Office of National Drug Control Policy. Epidemic: responding to America’s prescription drug abuse crisis. www.whitehouse.gov/sites/default/files/ondcp/issues-content/prescription-drugs/rx_abuse_plan.pdf. 2011. Accessed January 19, 2015.
- US Food and Drug Administration. Guidance for Industry: Abuse-Deterrent Opioids—Evaluation and Labeling. www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm334807.htm. Accessed January 23, 2015.
- Alexander L, Mannion RO, Weingarten B, et al. Development and impact of prescription opioid abuse deterrent formulation technologies. Drug Alcohol Depend. 2014;138:1-6.
- Schaeffer T. Abuse-deterrent formulations, an evolving technology against the abuse and misuse of opioid analgesics. J Med Toxicol. 2012;8:400-407.
- Stanos SP, Bruckenthal P, Barkin RL. Strategies to reduce the tampering and subsequent abuse of long-acting opioids: potential risks and benefits of formulations with physical or pharmacologic deterrents to tampering. Mayo Clin Proc. 2012;87:683-694.
- Cicero TJ, Ellis MS, Surratt HL. Effect of abuse-deterrent formulation of OxyContin. N Engl J Med. 2012;367:187-189.
- Butler SF, Cassidy TA, Chilcoat H, et al. Abuse rates and routes of administration of reformulated extended-release oxycodone: initial findings from a sentinel surveillance sample of individuals assessed for substance abuse treatment. J Pain. 2013;14:351-358.
- Cassidy TA, DasMahapatra P, Black RA, et al. Changes in prevalence of prescription opioid abuse after introduction of an abuse-deterrent opioid formulation. Pain Med. 2014;15:440-451.
- Coplan PM, Kale H, Sandstrom L, et al. Changes in oxycodone and heroin exposures in the National Poison Data System after introduction of extended-release oxycodone with abuse-deterrent characteristics. Pharmacoepidemiol Drug Saf. 2013;22:1274-1282.
- Severtson SG, Bartelson BB, Davis JM, et al. Reduced abuse, therapeutic errors, and diversion following reformulation of extended-release oxycodone in 2010. J Pain. 2013;14:1122-1130.
- Havens JR, Leukefeld CG, DeVeaugh-Geiss AM, et al. The impact of a reformulation of extended-release oxycodone designed to deter abuse in a sample of prescription opioid abusers. Drug Alcohol Depend. 2014;139:9-17.
- Michna E, Kirson NY, Shei A, et al. Use of prescription opioids with abuse-deterrent technology to address opioid abuse. Curr Med Res Opin. 2014;30:1589-1598.
- McNaughton EC, Coplan PM, Black RA, et al. Monitoring of Internet forums to evaluate reactions to the introduction of reformulated OxyContin to deter abuse. J Med Internet Res. 2014;16:e119.
- McNaughton EC, Black RA, Weber SE, Butler SF. Assessing abuse potential of new analgesic medications following market release: an evaluation of Internet discussion of tapentadol abuse. Pain Med. 2015;16:131-140.
